ARDS Just Got a Bedside Phenotyping Test. The Trial That Matters Hasn’t Run Yet.
PHIND demonstrates that hyperinflammatory and hypoinflammatory ARDS can be separated in under an hour at the bedside — but feasibility is not yet evidence of benefit, and the field needs to be careful about which question this paper actually answered.
For a decade, the hyperinflammatory and hypoinflammatory subphenotypes of ARDS have lived in the back rooms of retrospective trial datasets — biologically real, clinically suggestive, operationally inert. PHIND, published in Lancet Respiratory Medicine in March, is the first multicentre prospective study to drag that classification onto an actual ICU bedside in something close to a clinically actionable timeframe. The methodologic implications are larger than the trial itself, and they cut in both directions: PHIND validates a workflow, but it does not yet validate a therapy.
Why This Matters
ARDS has carried a sixty-year track record of pharmacologic null trials. The dominant explanation has shifted from “we have the wrong drugs” to “we are studying the wrong patient population” — specifically, that lumping biologically distinct ICU patients under one Berlin- or Global-definition umbrella dilutes any real treatment signal into a wash. If that explanation is correct, then every ARDS therapy worth re-examining — corticosteroids, neuromuscular blockade, conservative fluid strategy, simvastatin — depends on the field being able to identify subphenotypes at the bedside, prospectively, in time to enrol patients into trials and, eventually, into care pathways. PHIND is the operational proof that the door to that future is at least partially open. Whether it stays open depends on what happens in the prospective interventional trials that should follow this paper, not on this paper alone.
Study Design and Scope
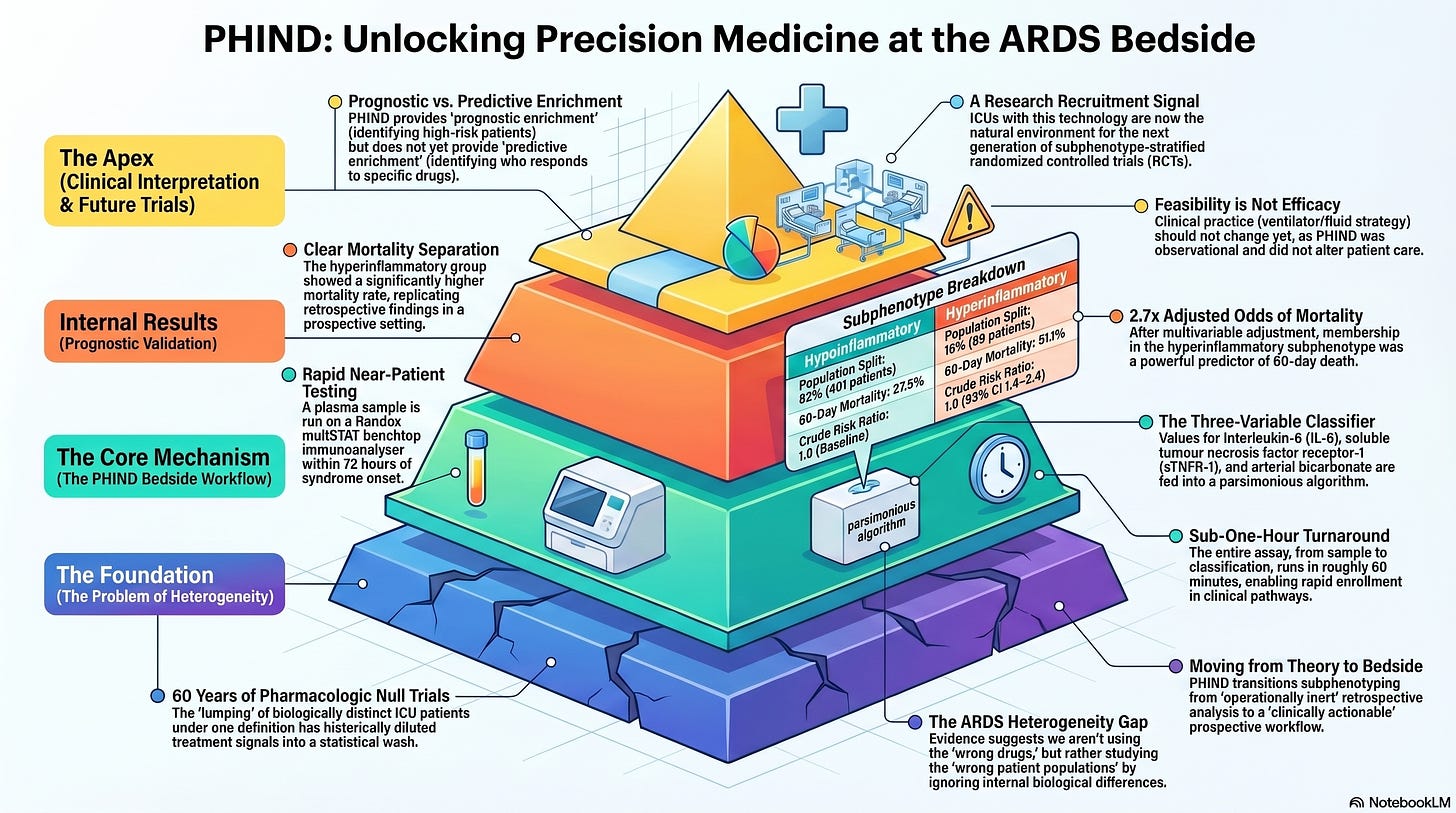
PHIND ran across thirty intensive care units within the NHS in the United Kingdom and the HSE in Ireland. Eligible adults carried a diagnosis of ARDS or acute hypoxaemic respiratory failure and were enrolled within seventy-two hours of syndrome onset. At enrolment, plasma was drawn and run on a near-patient benchtop immunoanalyser — the Randox Evidence multiSTAT — for two cytokines: interleukin-6 and soluble tumour necrosis factor receptor-1. Those values were fed, alongside arterial bicarbonate, into a previously validated parsimonious classifier that returns a probability of hyperinflammatory phenotype membership; a threshold of 0.5 separated the two groups. The whole assay, sample to classification, runs in roughly one hour. Five hundred twelve patients were enrolled, and four hundred ninety (95.7%) generated a usable classification. The primary outcome was sixty-day mortality. The trial is registered as NCT04009330.
What Stood Out Most
Three numbers carry the paper. First, distribution: eighteen percent of classified patients (89 of 490) fell into the hyperinflammatory subphenotype and eighty-two percent (401 of 490) into the hypoinflammatory subphenotype — a split that runs hyperinflammatory-low relative to most retrospective cohorts and worth flagging. Second, mortality: the hyperinflammatory group died at 51.1% by day sixty, versus 27.9% in the hypoinflammatory group, with a crude risk ratio of 1.8 (95% CI 1.4–2.4, p < 0.0001). Third, the adjusted signal: after multivariable adjustment, hyperinflammatory phenotype membership carried an odds ratio of 2.7 (95% CI 1.6–4.4) for sixty-day mortality. The prognostic separation replicated the retrospective literature with a tightness that is harder to dismiss when generated prospectively than when generated by latent class analysis of frozen trial samples a decade after the fact.
Interpretation
This paper is best read as a feasibility study with a strong prognostic-validation secondary endpoint, and not as a step toward changing care tomorrow morning. The contribution is real: a sub-one-hour bedside assay that classifies subphenotype with operationally meaningful fidelity, deployed across thirty ICUs in two health systems, is the missing infrastructure piece that has been blocking the next generation of ARDS trials. The contribution is also bounded. PHIND does not show that any therapy works differently across the two subphenotypes — it cannot, because it is observational. PHIND does not show that classifying patients changes outcomes — it cannot, because nothing about classification altered care. What it shows is that the door is unlocked for trials that test those questions directly, and the field should walk through that door rather than misreading PHIND as evidence that subphenotype-targeted therapy is already in hand.
The smaller and more dispersed point worth registering: the eighteen-percent hyperinflammatory prevalence is lower than the twenty-six to forty percent range typical of the original retrospective cohorts. Several explanations are plausible — different inclusion criteria, NHS and HSE case-mix differences, evolving baseline ARDS care, the inclusion of AHRF without Berlin-defined ARDS — and the paper does not adjudicate among them. Subscribers running journal clubs on this paper should not let that asymmetry pass without a discussion.
Watch the following video:
Bedside / Workplace Takeaways
Do not change ventilator strategy, fluid strategy, or steroid dosing on the basis of subphenotype today. No prospective interventional trial has yet shown that subphenotype-directed therapy changes outcomes. PHIND establishes feasibility, not efficacy. Anyone reading this paper as a directive for therapy is reading further than the data permit.
Update your mental model of “ARDS is heterogeneous.” That phrase has been an academic abstraction for a decade. It is now a clinical workflow with a one-hour turnaround. The bedside identification problem is solved at the engineering layer; what remains is the trial design problem.
For research-active units, this paper is a recruitment signal. Sites that ran PHIND and sites with comparable analyser access are the natural enrolment centres for the subphenotype-stratified trials that will follow. Fellowship programs with a clinical-trials track should be reading this paper as a roadmap to the next half-decade of ARDS RCT design.
For interprofessional teams, treat this as a methodology literacy moment. RTs running the ventilator on a hyperinflammatory patient and on a hypoinflammatory patient are running the same ventilator in two different biological diseases — until or unless prospective trials say otherwise. Naming that uncertainty in bedside rounds, rather than glossing over it, is the educated reading of the current evidence.
Do not let “near-patient assay” become shorthand for “point-of-care.” A benchtop immunoanalyser sitting in a unit-adjacent lab is operationally different from a true point-of-care cartridge at the bedside. The distinction matters for procurement, for sample handling, and for any subsequent regulatory pathway.
Teaching Pearl
This is the paper to use when teaching the difference between prognostic enrichment and predictive enrichment at journal club. Prognostic enrichment selects a population at higher risk of the outcome of interest, increasing the statistical power of a trial without requiring that the population respond differently to the intervention. Predictive enrichment selects a population biologically likelier to respond to the intervention. PHIND is a prognostic-enrichment paper: it shows that the hyperinflammatory group has higher mortality, which is exactly the prognostic signal you would design a trial around. The predictive-enrichment question — does this group respond differently to a specific therapy — is the question PHIND does not, and structurally cannot, answer. Fellowship learners who internalize this distinction will read the rest of the precision-critical-care literature more accurately.
Listen to the following podcast:
Over-Assumptions
The first over-assumption to watch for is the slide from feasibility to efficacy. A bedside classifier that works does not entail that classification-guided therapy works; the latter requires its own RCT. The second over-assumption is treatment-effect modification by analogy. Retrospective signals of differential response to fluid strategy, simvastatin, and corticosteroids exist in the literature, but none have been confirmed in prospective subphenotype-stratified trials, and informal extrapolation to standard-of-care decisions is a category error. The third is methodological: a parsimonious three-variable classifier is a calibrated abstraction of a richer biological state, and clinical readers should not mistake the classifier output for the underlying biology it approximates.
Limitations
The paper is observational and single-time-point; subphenotype membership at enrolment may not be stable over the course of an ICU admission, and recent work suggests meaningful transitions between phenotypes within the first thirty days. Generalizability beyond the NHS and HSE, and beyond the operational window in which a Randox multiSTAT analyser is unit-accessible, is an empirical question this work cannot resolve. The shifted prevalence relative to retrospective cohorts deserves scrutiny rather than assumption. And the classifier itself, although prospectively deployed, was derived retrospectively and inherits whatever biases lived in its training data.
Bottom Line
PHIND is a feasibility and prognostic-validation milestone for ARDS precision medicine: bedside subphenotype identification can be done in under an hour across thirty real-world ICUs, with mortality separation that mirrors the retrospective literature. It is not yet a license to change therapy. The next paper that matters is the subphenotype-stratified RCT.
Citation
Reddy K, Sinha P, Antcliffe DB, McDowell C, Bradley PA, Black L, et al; PHIND Investigators. Bedside identification of subphenotypes in acute respiratory failure (PHIND): a multicentre, observational cohort study. Lancet Respir Med. 2026. doi:10.1016/S2213-2600(26)00040-8
Calfee CS, Delucchi K, Parsons PE, Thompson BT, Ware LB, Matthay MA; NHLBI ARDS Network. Subphenotypes in acute respiratory distress syndrome: latent class analysis of data from two randomised controlled trials. Lancet Respir Med. 2014;2(8):611-620. doi:10.1016/S2213-2600(14)70097-9
Sinha P, Delucchi KL, McAuley DF, O’Kane CM, Matthay MA, Calfee CS. Development and validation of parsimonious algorithms to classify acute respiratory distress syndrome phenotypes: a secondary analysis of randomised controlled trials. Lancet Respir Med. 2020;8(3):247-257. doi:10.1016/S2213-2600(19)30369-8
Famous KR, Delucchi K, Ware LB, Kangelaris KN, Liu KD, Thompson BT, et al. Acute respiratory distress syndrome subphenotypes respond differently to randomized fluid management strategy. Am J Respir Crit Care Med. 2017;195(3):331-338. doi:10.1164/rccm.201603-0645OC
Calfee CS, Delucchi KL, Sinha P, Matthay MA, Hackett J, Shankar-Hari M, et al; Irish Critical Care Trials Group. Acute respiratory distress syndrome subphenotypes and differential response to simvastatin: secondary analysis of a randomised controlled trial. Lancet Respir Med. 2018;6(9):691-698. doi:10.1016/S2213-2600(18)30177-2
Matthay MA, Arabi Y, Arroliga AC, Bernard G, Bersten AD, Brochard LJ, et al. A New Global Definition of Acute Respiratory Distress Syndrome. Am J Respir Crit Care Med. 2024;209(1):37-47. doi:10.1164/rccm.202303-0558WS
⚠️ Medical Disclaimer: The content published in ICCN is intended solely for educational and informational purposes for healthcare professionals. It does not constitute medical advice, clinical guidelines, or a standard of care, and should not be used as a substitute for the independent professional judgment of a licensed clinician. All clinical decisions must be individualized to the patient and made by qualified healthcare providers. ICCN assumes no liability for any clinical outcomes arising from the information presented herein.
© 2026 Interprofessional Critical Care Network (ICCN). All rights reserved. Unauthorized reproduction or redistribution of this content is prohibited. Subscribers may share excerpts with proper attribution to ICCN and the author.