Reverse Triggering in 2026: Stop Asking If It's "Bad." Start Asking Which Phenotype You're Looking At.
A 2025 meta-analysis suggested it might be protective. A decade of physiology says it can pulverize the lung. Both are right — and the bedside answer is more demanding than either claim.
A patient on assist-control, deeply sedated, post-intubation day two for moderate ARDS. Their set rate is 22. Their actual delivered rate is 22. The waveform is metronome-clean. And every other breath, the esophageal manometer is showing a 12 cm H₂O negative deflection that nobody at the bedside is talking about, because the airway pressure tracing looks fine and the SpO₂ is 94%.
That patient is reverse triggering. Whether that fact is helping them or hurting them is one of the most important unresolved questions in modern mechanical ventilation — and the answer the field has converged on in 2026 is more interesting than the one it had in 2018.
Why This Matters
Reverse triggering is no longer a curiosity from a single 2013 case series. It is a routine, often-missed bedside event in 30–55% of sedated patients on controlled or assist-control ventilation, and the data linking it to outcomes have moved in two directions at once.¹ Physiologic studies show it can drive injurious pendelluft, breath stacking, and transpulmonary pressure swings that obliterate any benefit of low-tidal-volume ventilation.²,³,⁴ At the same time, a 2025 systematic review in Critical Care Medicine found that — unlike ineffective triggering and double triggering, which tracked with prolonged ventilation and longer ICU stays — reverse triggering was associated with a potentially favorable signal.⁵ Both readings are defensible. The field is not confused; the phenomenon is heterogeneous, and the clinical implication depends on which phenotype is in the room with you. ICUs that treat reverse triggering as a single entity are getting one of two equally bad answers half the time.
The Core Argument
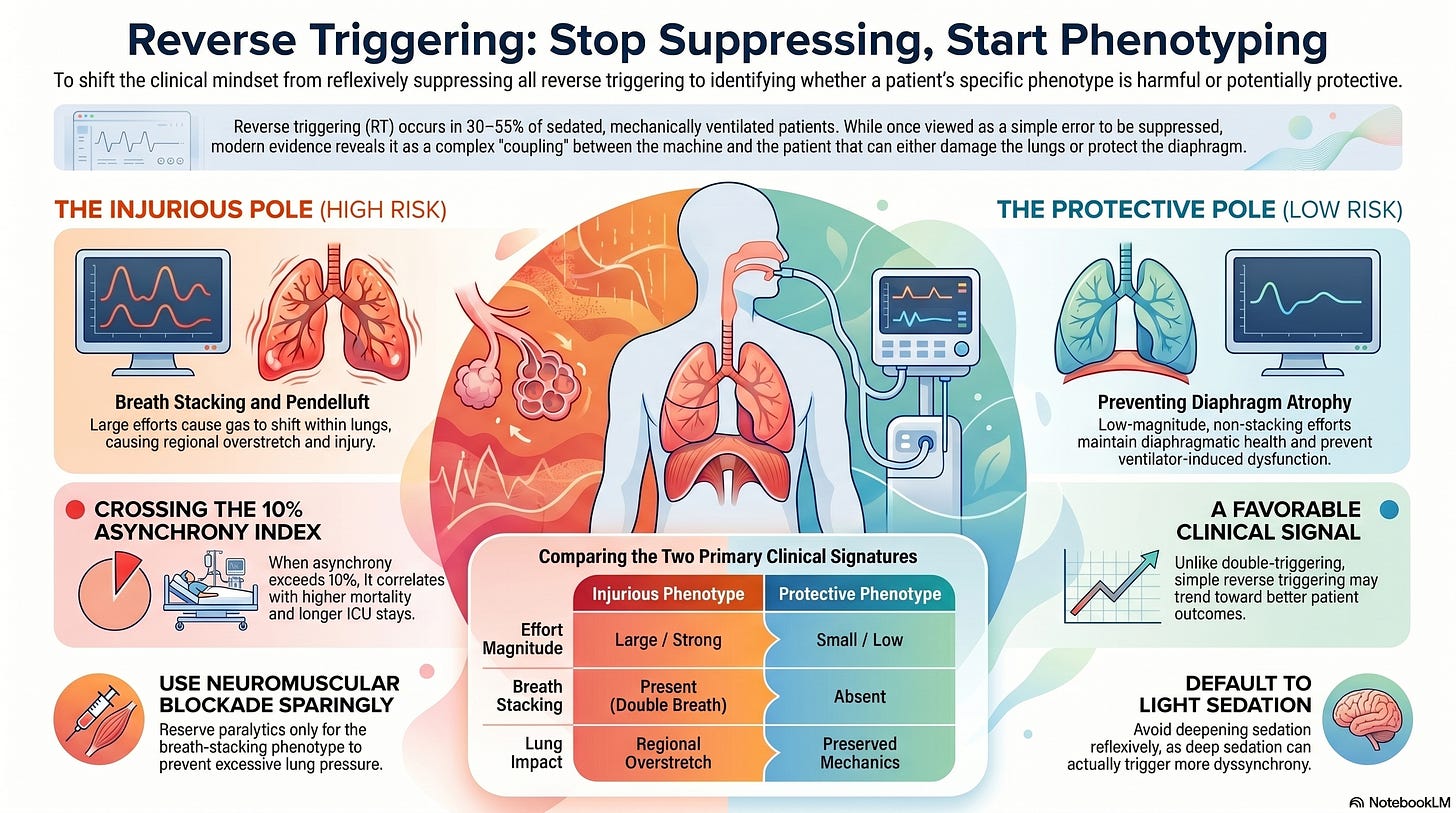
The right question is no longer “is reverse triggering harmful?” The right question is “which reverse trigger am I looking at, and what does the diaphragm-lung tradeoff actually look like in this patient, right now?” Reverse triggering is a phenotype space, not a binary, and the bedside discipline that follows from accepting that is concrete, learnable, and within the reach of any well-equipped ICU — not just academic centers running esophageal manometry on every patient. Treating reverse triggering as monolithic — either as a uniform threat to be paralyzed away or as a benign curiosity safely ignored — is the failure mode the next decade of mechanical ventilation has to retire.
What the Evidence Shows
The phenotypes are real, and they have separable physiologic signatures. In a single-center cohort of 55 ARDS patients on pressure-regulated ventilation, Baedorf Kassis and colleagues delineated four reverse-trigger patterns based on whether the patient effort fell during inflation or deflation and whether it produced breath stacking.² The phenotypes differed in their effects on tidal volume and transpulmonary pressure. This is not a continuum collapsed onto a single label; it is four distinct events the field had been calling one thing.
The injurious pole is well-characterized and not in dispute. Yoshida and colleagues showed in both an ARDS patient and an experimental injury model that strong reverse-triggered effort generates pendelluft — early-inspiratory transfer of gas from non-dependent to dependent lung — with regional overstretch and tidal recruitment in the very lung units a low-tidal-volume strategy is meant to spare.³ Breath-stacking phenotypes carry their own signature: when reverse-triggered effort is large enough to launch a second machine breath, the cumulative tidal volume and transpulmonary pressure swing exceed every threshold lung-protective ventilation was designed to honor.⁴ Asynchrony as a class — not reverse triggering specifically — has been associated with longer ICU and hospital stays and higher mortality when the asynchrony index crosses 10%.⁶
The protective-pole signal is newer and less established but no longer dismissible. The 2025 Critical Care Medicine systematic review pooled 19 studies and 2,672 patients and found that overall asynchrony, ineffective triggering, and double triggering each tracked with longer mechanical ventilation (mean difference 3.29 days; 95% CI 0.13–6.44) and longer ICU stays (3.65 days; 95% CI 1.20–6.11), but reverse triggering specifically separated from the other forms and trended toward a favorable association with outcomes.⁵ Mechanistically, this is consistent with the lung- and diaphragm-protective ventilation framework Goligher and colleagues advanced in 2020: a diaphragm that is fully unloaded for days atrophies fast, and any process that maintains low-magnitude, well-timed diaphragmatic activity may attenuate ventilator-induced diaphragm dysfunction.⁷ Reverse triggering, when the effort is small and not stacking, is — at least in principle — exactly that kind of activity.
Interpretation
The evidence is not contradictory. It is two truthful readings of two different phenotypes that the literature is only now learning to distinguish. The injurious-pole studies sampled patients with large effort, breath stacking, and pendelluft; their findings hold for those patients. The 2025 meta-analytic signal pooled studies in which reverse triggering was identified at the population level without phenotyping; their findings hold for that population in aggregate, where low-magnitude, non-stacking events likely dominate. The error has been in treating either result as a verdict on the phenomenon.
A more honest synthesis: reverse triggering is best understood as a marker of recovering respiratory drive that is being mishandled by sedation depth, rate setting, and inspiratory time. Whether it benefits the patient depends entirely on whether the magnitude of the reverse-triggered effort is small enough to preserve the diaphragm without distorting lung mechanics. Once the effort crosses that threshold — once it produces stacked breaths or measurable pendelluft — protective becomes injurious in real time, often before the bedside team notices.
The clinical implication is uncomfortable for ICUs that depend on time-cycled airway pressure tracings as their primary monitoring strategy. You cannot phenotype reverse triggering without esophageal pressure, electrical activity of the diaphragm, or — at minimum — a disciplined visual analysis of expiratory flow patterns and tidal volume drift across breaths.⁸ The field’s tooling is uneven, and most units are flying without the instruments needed to make this call.
Listen to the following podcast:
Bedside / Workplace Application
Stop reflexively deepening sedation. When reverse triggering appears, the instinct in many units is to push propofol or add a benzodiazepine. The literature on sedation depth and dyssynchrony points the other way: deep sedation is itself a setup for reverse triggering by suppressing the central pattern generator’s normal interplay with afferent input. Light, analgesia-first sedation is the more defensible default.⁷,⁸
Look at expiratory flow before reaching for a paralytic. A reverse trigger that dies on the expiratory limb without distorting tidal volume is a different event from one that produces a second machine breath. The ventilator is showing you the difference if anyone is watching the screen with intent. RTs and bedside nurses are the disciplines most likely to catch this; structure your unit’s monitoring so that catching it is treated as part of the job, not as an academic interest.
Reserve neuromuscular blockade for the breath-stacking phenotype, not for the rhythm itself. Neuromuscular blockade resolves all reverse triggering by definition, but it also imposes the full cost of complete diaphragm unloading in a patient who may not need it. The decision should be driven by physiologic evidence of injurious effort — not by the visual pattern alone.
Get esophageal manometry into routine use for ARDS patients on controlled ventilation longer than 48 hours. The barriers are training and culture, not technology. Units that have made this routine — Beth Israel Deaconess, Toronto General, the Brochard group — are not doing something other ICUs cannot do; they are doing something other ICUs have not yet committed to doing. ICCN’s position is that this becomes standard of care for moderate-to-severe ARDS within the next five years.
Phenotype before you suppress. If the magnitude of reverse-triggered effort is small, the timing is consistent, and there is no breath stacking, the most defensible action may be to do nothing — or, more precisely, to plan an early transition to assisted ventilation rather than to suppress the very respiratory drive that signals readiness for it.
Teaching Pearl
Teach reverse triggering not as a single waveform pattern but as a coupling problem between the ventilator’s set rhythm and the patient’s recovering respiratory drive. The Hering–Breuer reflex, mechanoreceptor afferents, and brainstem entrainment to ventilator-imposed inflation rhythm are the substrate. The clinical event is the interaction. When fellows and senior RTs learn to see it that way, the management questions follow naturally — what is the strength of coupling, what is the magnitude of effort, what is the inspiratory time, what is the sedation depth — instead of collapsing into “should I paralyze.” Frame it for in-services that way: not as an asynchrony to detect, but as a coupling to manage.
Counterpoints and Caveats
The strongest counterargument to ICCN’s position is that the protective-pole signal in the 2025 meta-analysis is fragile. The constituent studies were observational, the asynchrony detection methods were heterogeneous, and reverse triggering was identified at the population level rather than phenotyped. Reasonable readers will argue that one favorable meta-analytic signal does not justify treating reverse triggering as anything other than an asynchrony to suppress. That argument is internally consistent and not unreasonable.
A second counter, advanced credibly by methodologists in this space, is that the pendelluft/breath-stacking phenotype is so injurious — and so under-detected without esophageal pressure — that defaulting to suppression is the safer policy precisely because units cannot reliably tell the phenotypes apart. The risk-tolerance argument is not nothing. ICCN’s response is that policy that flattens phenotypes always produces avoidable harm at one of the poles; the right answer is to invest in the monitoring infrastructure that makes phenotyping routine, not to choose a one-size answer because the infrastructure is currently uneven.
Watch the following video explainer:
Limitations of the Argument
The phenotype-first framework presumes monitoring capacity that many community ICUs do not have. In units without esophageal manometry, EAdi catheters, or high-fidelity expiratory flow analysis, the argument’s bedside utility shrinks. A reasonable interim default in those settings is closer to the suppression posture — not because it is correct, but because the cost of missed injurious phenotypes is greater than the cost of over-suppressed protective ones. The argument also rests on a 2025 meta-analytic signal that is hypothesis-generating rather than confirmatory; ongoing trials phenotyping reverse triggering against ventilatory adjustments will likely reshape the picture within two to three years. Finally, none of this applies to patients in the most acute phase of severe ARDS where the lung-protective imperative dominates every other consideration; in that population, neuromuscular blockade for the first 48 hours remains defensible regardless of phenotype.
Bottom Line
Reverse triggering is not a single thing. The injurious phenotype demands intervention; the low-magnitude, non-stacking phenotype may be doing diaphragm work the unit otherwise has to engineer in. The clinical skill is telling them apart, and the ICUs that build that skill — through esophageal manometry, disciplined waveform reading, and an interprofessional culture in which RTs and bedside nurses are trusted to flag these events — will own this problem in 2026. Everyone else will keep guessing.
References
Akoumianaki E, Lyazidi A, Rey N, et al. Mechanical ventilation-induced reverse-triggered breaths: a frequently unrecognized form of neuromechanical coupling. Chest. 2013;143(4):927-938. doi:10.1378/chest.12-1817
Baedorf Kassis E, Su HK, Graham AR, Novack V, Loring SH, Talmor DS. Reverse trigger phenotypes in acute respiratory distress syndrome. Am J Respir Crit Care Med. 2021;203(1):67-77. doi:10.1164/rccm.201907-1427OC
Yoshida T, Nakamura MAM, Morais CCA, Amato MBP, Kavanagh BP. Reverse triggering causes an injurious inflation pattern during mechanical ventilation. Am J Respir Crit Care Med. 2018;198(8):1096-1099. doi:10.1164/rccm.201804-0649LE
Beitler JR, Sands SA, Loring SH, et al. Quantifying unintended exposure to high tidal volumes from breath stacking dyssynchrony in ARDS: the BREATHE criteria. Intensive Care Med. 2016;42(9):1427-1436. doi:10.1007/s00134-016-4423-3
de Bie MJ, Rietveld PJ, et al. The Association Between Patient-Ventilator Asynchrony and Clinical Outcomes in Mechanically Ventilated Patients: A Systematic Review. Crit Care Med. 2025;53(11):e2261-e2270. doi:10.1097/CCM.0000000000006816
Blanch L, Villagra A, Sales B, et al. Asynchronies during mechanical ventilation are associated with mortality. Intensive Care Med. 2015;41(4):633-641. doi:10.1007/s00134-015-3692-6
Goligher EC, Dres M, Patel BK, et al. Lung- and diaphragm-protective ventilation. Am J Respir Crit Care Med. 2020;202(7):950-961. doi:10.1164/rccm.202003-0655CP
Rodrigues A, Telias I, Damiani LF, Brochard L. Reverse triggering during controlled ventilation: from physiology to clinical management. Am J Respir Crit Care Med. 2023;207(5):533-543. doi:10.1164/rccm.202208-1477CI